 VOLUME 27, ISSUE 2 • JUNE 2023 Full issue »
VOLUME 27, ISSUE 2 • JUNE 2023 Full issue »

Motor differences between multiple system atrophy and Parkinson's disease: The key is also in sleep
Sleep plays an important diagnostic and prognostic role among α-synuclein neurodegeneration. As sleep disorders are common features of multiple system atrophy and Parkinson's disease, the higher prevalence of prominent motor dyscontrol during sleep (Fig. 1) and REM sleep behavior disorder (RBD) in MSA may be a cue for differential diagnosis. Indeed tonic electromyographic (EMG) activity of submentalis and tibialis anterior muscles was reported as common in subjects with MSA compared to those with obstructive sleep apneas, although without quantitative confirmation1.
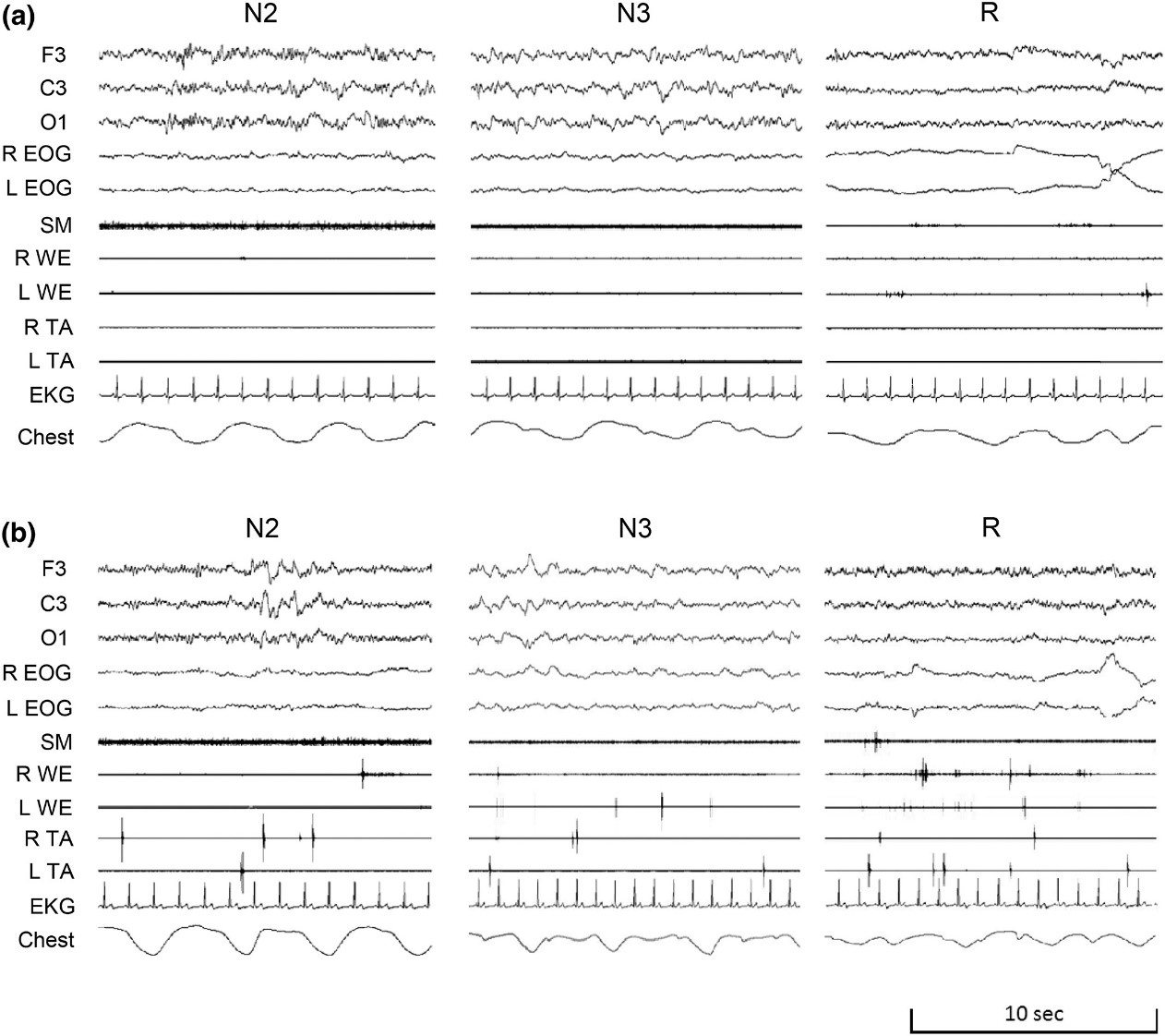
Figure 1. The panel shows representative polysomnographic recordings in a subject with Parkinson's disease (panel a) and one with multiple system atrophy (panel b) showing typical diffuse and pervasive muscular activation spanning through both non-REM and REM sleep and different localization (axial and limbs). The tracings include the electroencephalogram (F3, C3, O1 leads), right (R) and left (L) electrooculogram (EOG), the submentalis muscle (SM), the R and L wrist extensor muscle (WE), and the R and L tibialis anterior muscle (TA) electromyograms, the electrocardiogram (EKG), and the thoracic respirogram (chest) during stages N2, N3, and R sleep.
In a recently published paper in Journal of Sleep Research entitled “Pervasive and diffuse muscle activity during REM sleep and non-REM sleep characterizes multiple system atrophy in comparison with Parkinson's disease,”2 we applied a novel automatic EMG analysis technique3 based on distributions of normalized EMG values (DNE) to investigate whether pervasive muscle activity during non-REM sleep and REM sleep occurred in different muscles in subjects with MSA vs. PD.
To test our hypotheses, we included the first 50 consecutive subjects with PD enrolled in the BoProPark study4 and 26 age- and gender-matched subjects with MSA at <5 years from disease onset. The DNE analysis focused on submentalis and on bilateral wrist extensor and tibialis anterior muscles in different wake-sleep states during the night.
Subjects with MSA had significantly higher activity of submentalis, wrist extensor, and tibialis anterior muscles than subjects with PD during non-REM sleep, including separately in stages N1, N2, and N3, and during REM sleep, but not during nocturnal wakefulness (Fig. 2). Moreover, DNE indexes of EMG activity of wrist extensor and tibialis anterior muscles during non-REM sleep were significantly higher in subjects with MSA and RBD than in those with PD with RBD. The EMG summary index values did not correlate significantly with LEDD, Hoehn-Yahr score, or UPDRS III score. However, in subjects with MSA, DNE indexes of EMG activity of submentalis muscles during sleep correlated positively and significantly with disease duration.
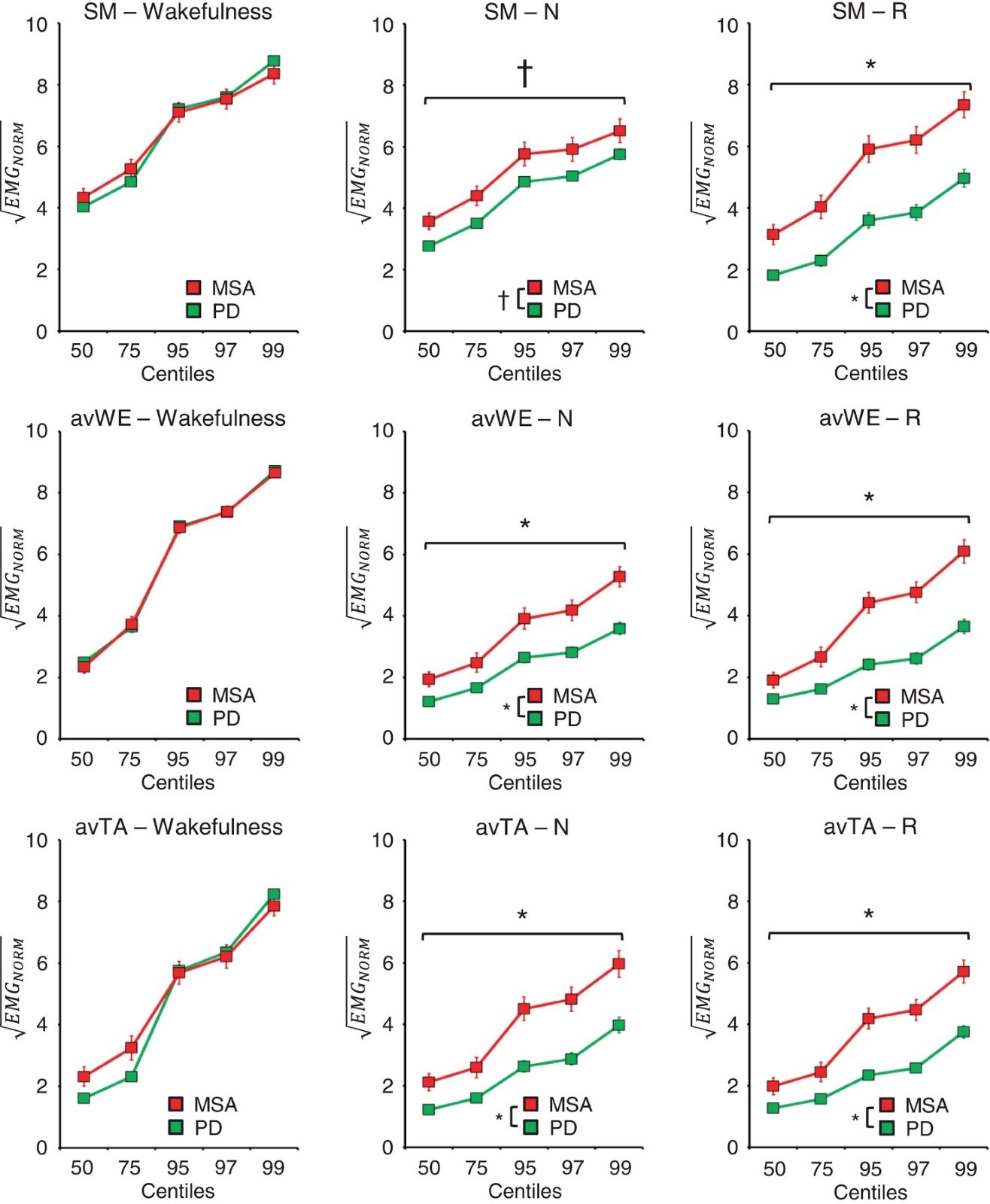
Figure 2. The panels show the distributions of normalized electromyographic activity (EMGNORM) of submentalis (SM), average left and right wrist extensor (avWE), and average left and right tibialis anterior (avTA) muscles during wakefulness, stages N1, N2, and N3 sleep combined (N), and stage R sleep in subjects with Parkinson's disease (PD) or multiple system atrophy (MSA). *p < 0.05, PD versus MSA, t-test with false discovery rate correction referred to all centiles above horizontal bracket. †p < 0.05, 2-way ANOVA main effect in the absence of significant group x centile interaction.
Taken together, these analyses indicate that with respect to PD, MSA within 5 years from disease onset was characterized by a pervasive and diffuse muscle overactivity involving axial and limb muscles and occurring not only during REM sleep, but also during non-REM sleep and with comorbid RBD.
Our findings represent, to our knowledge, the first complete and simultaneous quantification of EMG activity of different muscles during non-REM sleep in subjects with PD and MSA. Previous work had mainly focused on the analysis of submentalis EMG activity during REM sleep, demonstrating a lack of muscle atonia in MSA5, also with respect to PD6.
Our findings may be linked to the different and more extensive pattern of neuropathology in MSA versus PD, as they are not solely accounted by the greater prevalence of RBD in MSA. These results may lay the basis for prognostic biomarkers and stress the interest of assessing muscle control not only during REM sleep, but also during non-REM sleep.
References
1. Vetrugno R, Provini F, Cortelli P, et al. Sleep disorders in multiple system atrophy: A correlative video-polysomnographic study. Sleep Med. 2004;5(1):21-30. doi:10.1016/j.sleep.2003.07.002
2. Silvani A, Baldelli L, Giannini G, et al. Pervasive and diffuse muscle activity during REM sleep and non-REM sleep characterises multiple system atrophy in comparison with Parkinson’s disease. J Sleep Res. 2022;n/a(n/a):e13721. doi:https://doi.org/10.1111/jsr.13721
3. Silvani A, Ferri R, Lo Martire V, et al. Muscle activity during sleep in human subjects, rats, and mice: Towards translational models of REM sleep without atonia. Sleep. 2017;40(4). doi:10.1093/sleep/zsx029
4. Calandra-Buonaura G, Sambati L, Baschieri F, et al. The Bologna motor and non-motor prospective study on parkinsonism at onset (BoProPark): study design and population. Neurol Sci. 2020;41(9):2531-2537. doi:10.1007/s10072-020-04305-9
5. Ferri R, Manconi M, Plazzi G, et al. A quantitative statistical analysis of the submentalis muscle EMG amplitude during sleep in normal controls and patients with REM sleep behavior disorder. J Sleep Res. 2008;17(1):89-100. doi:10.1111/j.1365-2869.2008.00631.x
6. Iranzo A, Santamaría J, Rye DB, et al. Characteristics of idiopathic REM sleep behavior disorder and that associated with MSA and PD. Neurology. 2005;65(2):247-252. doi:10.1212/01.wnl.0000168864.97813.e0
Read more Moving Along:






