 VOLUME 26, ISSUE 1 • March 2022. Full issue »
VOLUME 26, ISSUE 1 • March 2022. Full issue »

The Diagnostic Pathway of Imaging in Clinical Practice for Parkinsonisms
Share this:
Considerable progress has been made in the development of biomarkers for Parkinson's disease (PD) or atypical Parkinsonisms (APDs). In contrast with these advancements, their diagnosis remains principally based on clinical criteria. However, the new International Parkinson and Movement Disorder Society (MDS) diagnostic criteria for PD include the novelty of incorporating imaging as part of “absolute exclusion criteria” (normal presynaptic dopaminergic terminal scan) and “supportive criteria” (cardiac sympathetic denervation using (123)I metaiodobenzyl guanidine). (1) Similarly, the utility of cerebral glucose metabolism with [18F]FDG-PET has been suported by the MDS criteria for the diagnosis of progressive supranuclear palsy (PSP) and the latest criteria for the diagnosis of multiple system atrophy (MSA). (2, 3)
In a recently published paper in Movement Disorders Clinical Practice entitled “Pragmatic Approach on Neuroimaging Techniques for the Differential Diagnosis of Parkinsonisms” (4), we discussed how the neuroimaging field evolved to change the role of imaging from merely excluding secondary causes of Parkinsonisms to aiding in the differential diagnosis.
To contribute to narrow the gap between the increasing recognition of imaging techniques and the lack of imaging protocols or algorithms on how to use them in daily practice for differential diagnosis, we delineated a pragmatic approach to describe the basic steps that may not be missed in clinical practice. Essentially, the paper builds a picture of the connection between the imaging criteria that support the diagnosis of PD or APDs and their integration with clinical aspects, through 2 algorithms, one for MRI and another for PET/SPECT imaging (Fig 1).
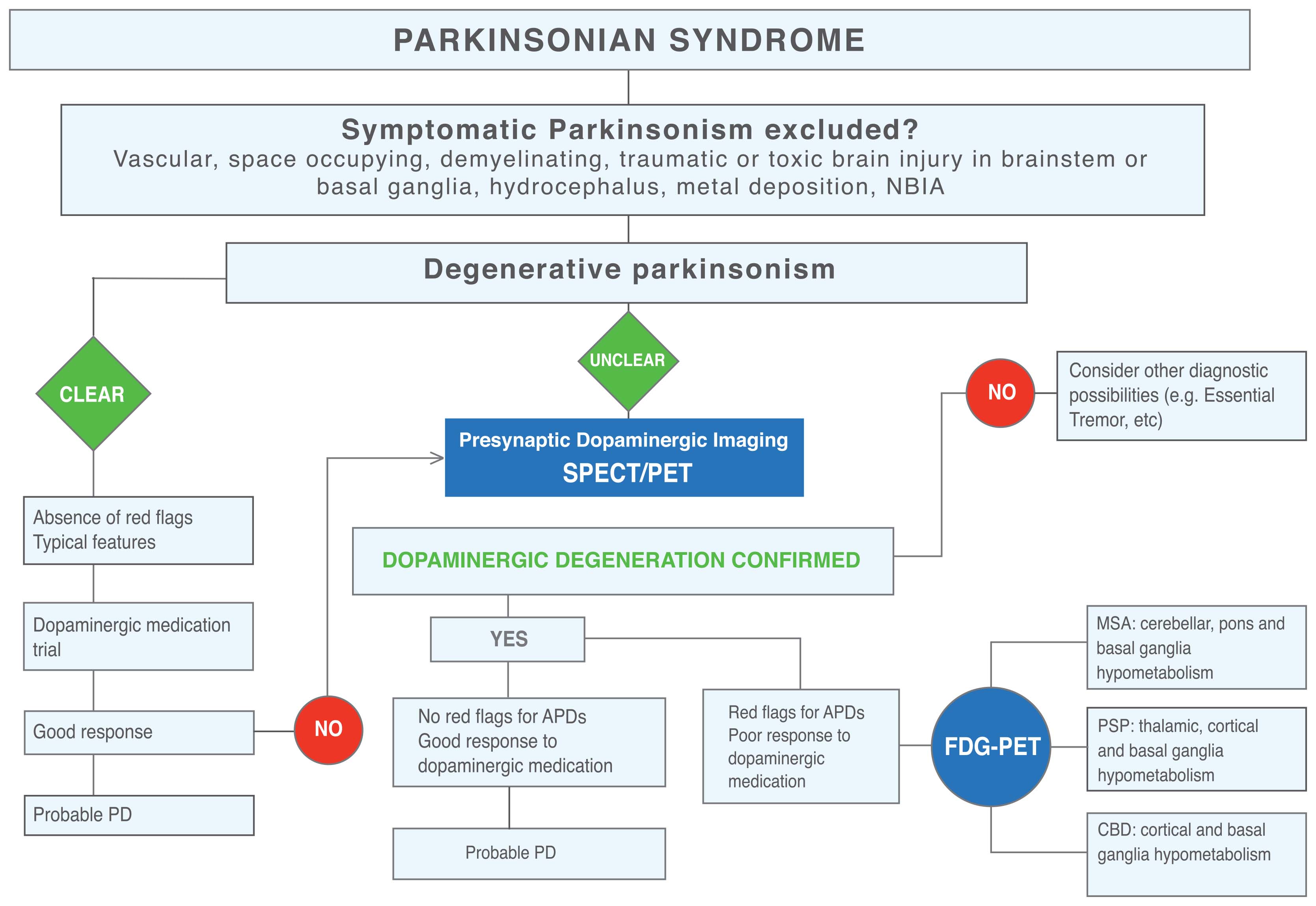
Today, high-field MR using qualitative techniques of Neuromelanin and Dorsal Nigral Hyperintensity imaging, allow the identification of the ongoing neurodegenerative process, the loss of dopaminergic cells and the decrease in neuromelanin signal, and Iron-sensitive MRI using susceptibility weighted imaging, allow the detection of a decreased signal in the putamen in patients with APDs, where the iron deposition is more prevalent and severe in PSP compared with MSA-P, except in the putamen that contains more iron in MSA-P. (5, 6)
The Molecular imaging field gives us the best opportunity to understand pathogenesis in Parkinsonisms and currently also provide the possibility of imaging protein aggregation. In this regard, a marker of the dopamine transporter, DAT-SPECT, was approved by FDA for the differential diagnosis of PD from Essential Tremor, although other markers for presynaptical dopaminergic function are used in clinical practice. [18F]FDG-PET studies are especially relevant because they identify “patterns of brain hypometabolism” and offer to the clinician a strong “imaging–clinical” correlation, which is particularly valuable for the diagnosis of neurodegenerative diseases.
Tau PET imaging is probably the most challenging area in Parkinsonisms that may evolve as an in vivo modality supportive of pathological PSP diagnosis at the individual level, as shown by second generation tracers such as[18F]-PI-2620, which demonstrated high in vitro binding affinity for aggregated 4-repeat isoforms and no substantial off-target binding to β-amyloid or MAO A or B, highlighting that patients with PSP-RS and non-RS presented significant elevation of tracer binding in PSP target regions. (7)
Share this: Tweet
References
1. Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord 2015;30(12):1591–1560.
2. Höglinger G, Respondek G, Stamelou M, et al. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord 2017;32(6):853–864
3. Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008;71(9): 670–676.
4. Peralta C, Strafella A, van Eimeren T, Ceravolo R, Seppi K, Kaasinen V, Arena J, Lehericy S, International Parkinson Movement Disorders Society-Neuroimaging Study Group. Pragmatic Approach on Neuroimaging Techniques for the Differential Diagnosis of Parkinsonisms. Mov Disord Clin Pract. 2021 Nov 15;9(1):6-19.
5. Ohtsuka C, Sasaki M, Konno K, et al. Changes in substantia nigra and locus coeruleus in patients with early-stage Parkinson’s disease using neuromelanin-sensitive MR imaging. Neurosci Lett 2013;541:93–98.
6. Mahlknecht P, Krismer F, Poewe W, Seppi K. Meta-analysis of dorsolateral Nigral Hyperintensity on magnetic resonance imaging as a marker for Parkinson’s disease. Mov Disord 2017;32(4):619–623.
7. Bullich S, Barret O, Constantinescu C, et al. Evaluation of Dosimetry, quantitative methods, and test-retest variability of 18 F-PI-2620 PET for the assessment of tau deposits in the human brain. J Nucl Med 2020;61(6): 920–927.
Read more Moving Along:






